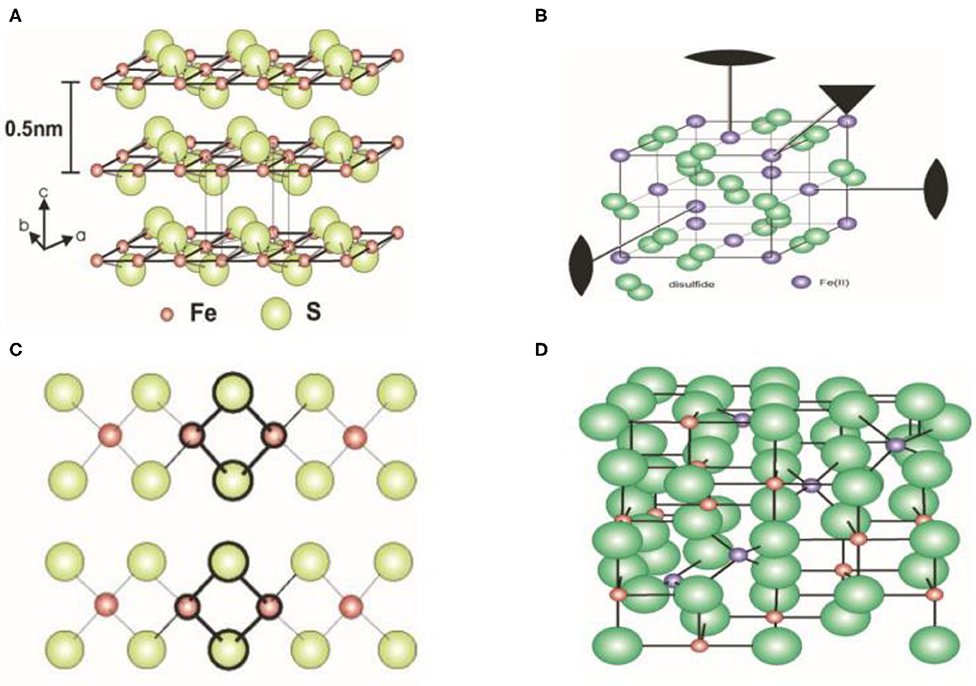
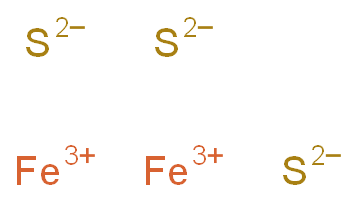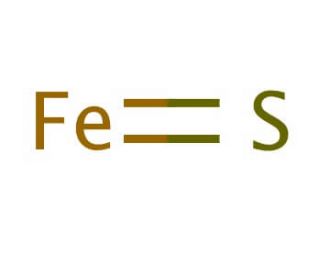SOLVED: Compound Formula Ksp Iron(II) sulfide FeS 3.72 X 10-19 Magnesium Mg(OH)2 2.06 X 10-13 hydroxide Calcium CaF2 1.46 X 10-10 fluoride Refer to the Ksp values in the table above to

Law of Conservation of Matter The Law of Conservation of Matter states that matter can neither be created nor destroyed.The Law of Conservation of Matter. - ppt download
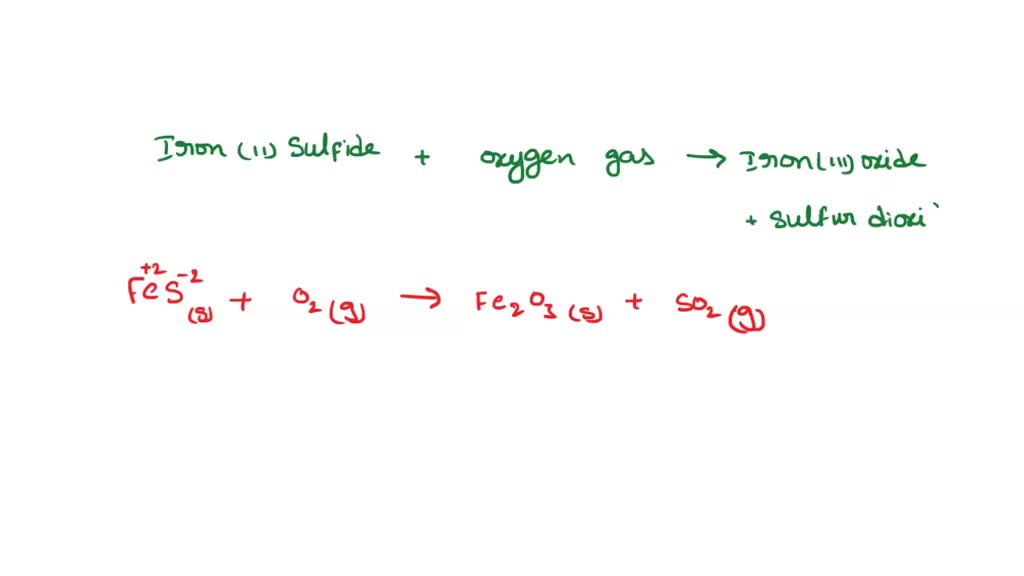
SOLVED: A student reacted iron (II) sulfide with oxygen gas to form iron (III) oxide and sulfur dioxide. What is the balanced equation? 4FeS(s) + 7O2(g) â†' 2Fe2O3(s) + 4SO2(g)

SOLVED:Iron(II) sulfide reacts with hydrochloric acid according to the reaction: FeS(s)+2 HCl(a q) ⟶FeCl2(s)+H2 S(g) A reaction mixture initially contains 0.223 mol Fes and 0.652 mol HCl. Once the reaction has occurred