Energies | Free Full-Text | Capacity Decay Mechanism of the LCO + NMC532/Graphite Cells Combined with Post-Mortem Technique

Effect of Electrolyte Additives on the LiNi0.5Mn0.3Co0.2O2 Surface Film Formation with Lithium and Graphite Negative Electrodes - Hekmatfar - 2020 - Advanced Materials Interfaces - Wiley Online Library
Upcycling Low-Nickel Polycrystalline Cathodes from Retired Electric Vehicle Batteries into Single-Crystal Nickel-Rich Cathodes G

Examining Effects of Negative to Positive Capacity Ratio in Three-Electrode Lithium-Ion Cells with Layered Oxide Cathode and Si Anode | ACS Applied Energy Materials
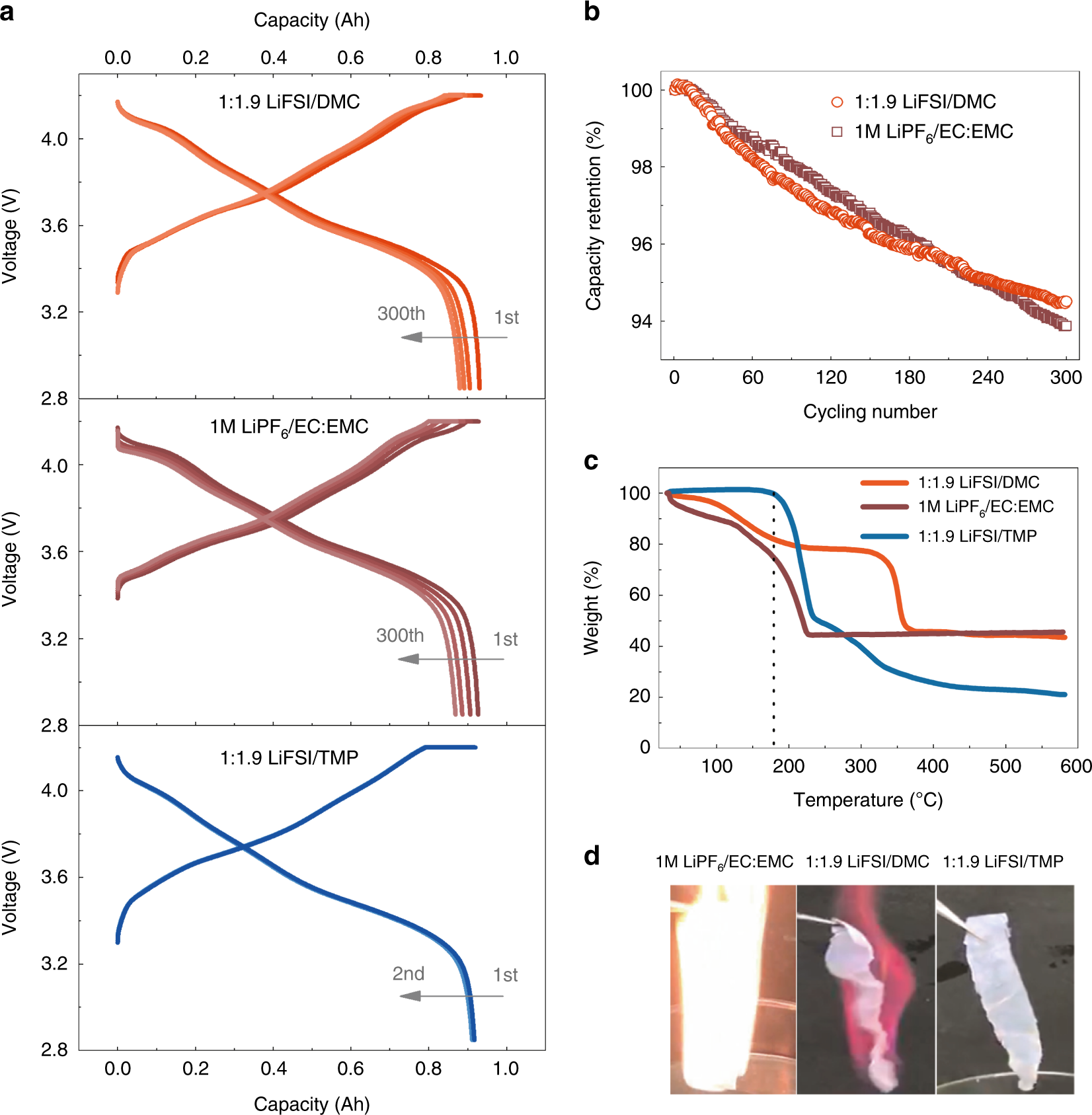
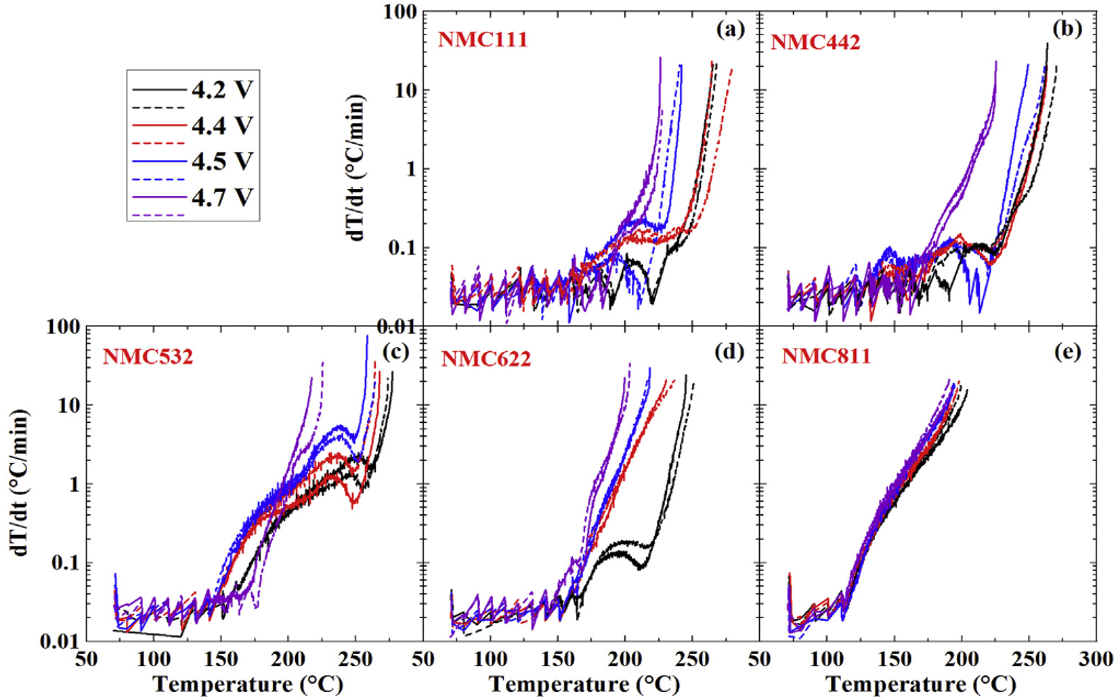
Thermal runaway of Lithium-ion batteries employing LiN(SO2F)2-based concentrated electrolytes | Nature Communications
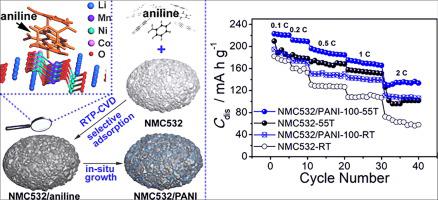
Selective adsorption-involved formation of NMC532/PANI microparticles with high ageing resistance and improved electrochemical performance,Journal of Energy Chemistry - X-MOL

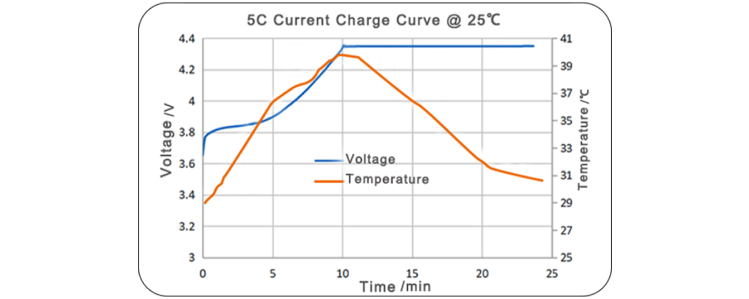
LiNiMnCoO2 NMC532 Lithium Nickel Manganese Cobalt Oxide NMC523 NCM523 Powder for Lithium ion Battery Cathode Material
Deciphering Interfacial Chemical and Electrochemical Reactions of Sulfide‐Based All‐Solid‐State Batteries
![PDF] Single crystal cathodes enabling high-performance all-solid-state lithium-ion batteries | Semantic Scholar PDF] Single crystal cathodes enabling high-performance all-solid-state lithium-ion batteries | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/04235a39133fd4da46e194712063da4a3a9d20b0/3-Figure3-1.png)
PDF] Single crystal cathodes enabling high-performance all-solid-state lithium-ion batteries | Semantic Scholar

EV Battery Component Maker Natrion Gets Closer to Solid-State Reality with High-Performance, Heat-Stable Multilayer Pouch Cells – Natrion

Electrochemical evaluation of LiNi0.5Mn0.3Co0.2O2, LiNi0.6Mn0.2Co0.2O2, and LiNi0.8Mn0.1Co0.1O2 cathode materials for lithium-ion batteries: from half-coin cell to pouch cell | SpringerLink
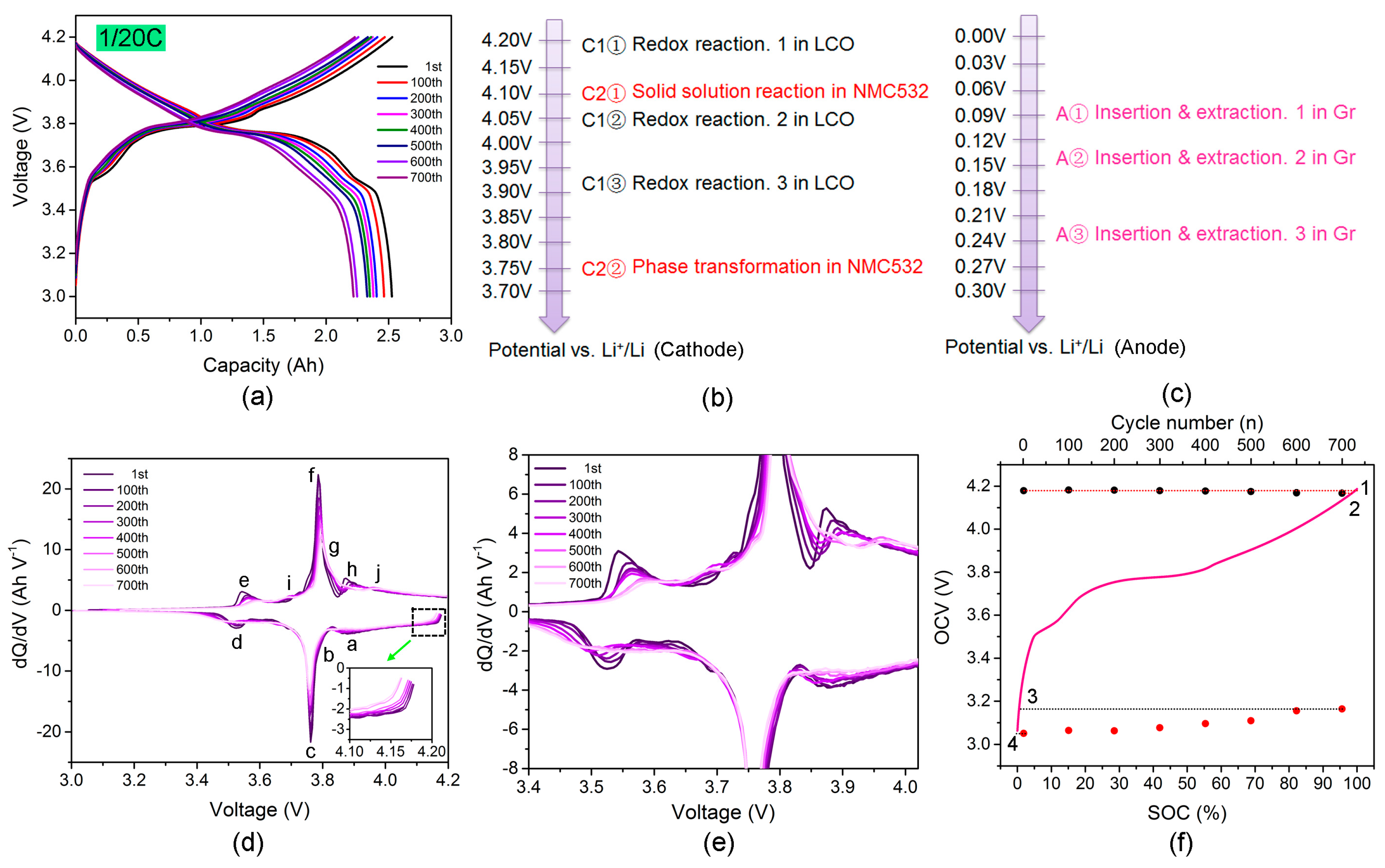
Energies | Free Full-Text | Capacity Decay Mechanism of the LCO + NMC532/Graphite Cells Combined with Post-Mortem Technique
Comparison of Single Crystal and Polycrystalline LiNi0.5Mn0.3Co0.2O2 Positive Electrode Materials for High Voltage Li-Ion Cells

Examining Effects of Negative to Positive Capacity Ratio in Three-Electrode Lithium-Ion Cells with Layered Oxide Cathode and Si Anode | ACS Applied Energy Materials

a) Anode-free pouch cell (NMC532/Cu) used in this work (left) compared... | Download Scientific Diagram

Selective adsorption-involved formation of NMC532/PANI microparticles with high ageing resistance and improved electrochemical performance - ScienceDirect